First Cannabis-Derived Rx Drug Now On The Market
Written by Chris Weatherall on Jan 9, 2019
Medical marijuana laws have changed dramatically across the country in recent years, and now cannabis pharmaceuticals appear to be the next big leap forward in cannabis law reform. While many critics of medical cannabis continue to debate legalization of cannabis pros and cons, the reality is that the new FDA-approved cannabis drug could reshape how pharmaceutical companies view medical cannabis and encourage them to explore new consumer medical applications of cannabis.
Medical Marijuana In Pill Form, With FDA Approval
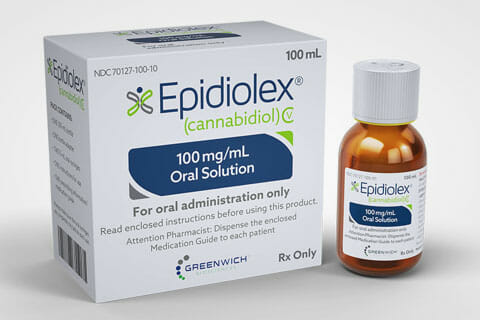 The first-ever cannabis-based drug with FDA approval is Epidihttps://kindmedsaz.com/wp-content/uploads/2019/01/First-Cannabis-Derived-Rx-Drug-Now-On-The-Market.jpgolex*, a new prescription drug designed to treat severe seizure disorders that do not respond to other treatments. The Epidiolex FDA approval is big news for the medical marijuana industry and could help improve perceptions about medical cannabis all over the country. After securing FDA approval in June of 2018, Epidiolex’s manufacturer GW Pharma announced the drug should enter the consumer market by the end of 2018.
The first-ever cannabis-based drug with FDA approval is Epidihttps://kindmedsaz.com/wp-content/uploads/2019/01/First-Cannabis-Derived-Rx-Drug-Now-On-The-Market.jpgolex*, a new prescription drug designed to treat severe seizure disorders that do not respond to other treatments. The Epidiolex FDA approval is big news for the medical marijuana industry and could help improve perceptions about medical cannabis all over the country. After securing FDA approval in June of 2018, Epidiolex’s manufacturer GW Pharma announced the drug should enter the consumer market by the end of 2018.
Cannabis Legalization Throughout The Country
The recent years have seen drastic marijuana law changes in several states. More than half the states in the country have now legalized medical cannabis for patients with qualifying conditions and several more have gone so far as to legalize recreational marijuana for adults. There has been substantial cannabis legalization news lately, and Epidiolex’s entrance into the consumer pharmaceutical market is one of the most important stories.
Read More: Arizona Cannabis Legalization & Medical Marijuana
What Does Epidiolex Treat?
GW Pharma produced Epidiolex to meet a demand for alternative seizure disorder treatment, particularly for children who suffer from childhood seizure disorders that are unresponsive to conventional treatments. GW Pharma carefully developed Epidiolex using cannabinoids to reduce the occurrence of seizures. Thanks to their diligence in perfecting the drug and ensuring its safety for intended use, the Drug Enforcement Agency scheduled Epidiolex under the lowest Schedule classification for controlled substances.
What You Should Know About Epidiolex
In plant form, marijuana still falls under Schedule I classification alongside heroin. However, the entrance of Epidiolex into the consumer market bolsters the evidence that marijuana does have medical value. Schedule I classification typically pertains to narcotic substances with no medical value. Marijuana law reform advocates and many medical researchers have been fighting to change marijuana’s Schedule I classification for years. Epidiolex may offer them more evidence to prove marijuana’s medical value and encourage adjustment of current marijuana scheduling at the federal level.
Epidiolex has received FDA approval to treat Dravet Syndrome and Lennox-Gestaut Syndrome; two very rare forms of epilepsy that manifest in early childhood. Children who suffer from these conditions generally experience social and developmental delays due to constant seizures. Epidiolex contains no THC, the main psychoactive ingredient that produces euphoria from smoking marijuana. Instead, Epidiolex contains refined cannabidiol (CBD), a compound that provides pain relief and anti-seizure properties without the typical effects associated with marijuana use.
Dravet Syndrome
This seizure disorder typically appears in infancy and can cause febrile seizures that result in high fever. Children with this condition later develop spastic or myoclonic seizures which can cause involuntary muscle movements. Repeat seizures can result in a life-threatening condition called status epilepticus that requires emergency medical treatment. Children born with Dravet syndrome generally have developmental delays, social impairment, hyperactivity, and delayed motor skill development.
Lennox-Gestaut Syndrome
This seizure disorder generally manifests between the ages of three and five. Children with this condition will experience different types of seizures, but the most common for Lennox-Gestaut are tonic seizures that cause involuntary muscle contractions. Children born with Lennox-Gestaut syndrome often have intellectual impairment, delayed development, poor motor skills, and they may require long-term in-home care.
These types of seizure disorders have a very high chance of causing long-term or permanent damage in many ways. Effective treatment isn’t always possible, and many of the children born with these seizure disorders require constant in-home care. The only side effects reported from Epidiolex clinical trials were sleepiness, sedation, insomnia, fatigue, diarrhea, rash, and some infections. However, these side effects pale in comparison to the relief Epidiolex can offer children struggling with advanced seizure disorders.
Growing Acceptance For Cannabis As Medicine
 State law changes in recent years indicate a growing acceptance of medical cannabis across the country. A recent study from the Pew Research Center** reported that 62% of American adults support full legalization of marijuana for recreational use for adults, and the amount of support for medical marijuana is even higher. While some people are still hesitant to support or advocate recreational marijuana legalization, they may still acknowledge the medical value of cannabis for patients across the country.
State law changes in recent years indicate a growing acceptance of medical cannabis across the country. A recent study from the Pew Research Center** reported that 62% of American adults support full legalization of marijuana for recreational use for adults, and the amount of support for medical marijuana is even higher. While some people are still hesitant to support or advocate recreational marijuana legalization, they may still acknowledge the medical value of cannabis for patients across the country.
Even conservatives who have traditionally remained staunchly opposed to any type of marijuana legalization are now reporting more support for marijuana law reform than ever before. New marijuana treatment options like prescription drugs may help change even more minds due to the common perception of prescription drugs as safe. Another potentially valuable benefit of more widespread acceptance of cannabis-based medications is the replacement of prescription opioid painkillers, the driving force behind the opioid crisis that has enveloped the United States for over a decade.
Cannabis For A Broader Consumer Market
Medical marijuana has evolved tremendously in recent years. Dried flower is no longer the only option. There are topical creams, tinctures, distillates, concentrates, edible products, and now prescription medications that use cannabis as a healing ingredient. This level of variety is necessary because while marijuana can be an effective treatment for several medical conditions, not every patient can consume cannabis the traditional way by smoking it.
For example, medical marijuana can greatly ease the stress of cancer treatment. It can help a patient’s nausea from chemotherapy, encourage a healthy appetite, and provide relief from the stress that cancer and treatment cause. However, a person suffering with lung cancer should not smoke anything, even cannabis with healing properties. Instead, edible products, topical creams, or other medical cannabis products can offer relief without aggravating the patient’s original condition.
Medical Marijuana Benefits Without The High
One of the major factors behind the FDA approval of Epidiolex is the fact that the drug only contains CBD and has a 0% THC concentration. Since the psychoactive effects of marijuana may not appeal to some people with qualifying conditions, CBD-based medications can be a valuable addition to the consumer pharmaceutical market. CBD medications like Epidiolex are especially beneficial for children who should not consume THC. Parents of children who suffer from Dravet syndrome, Lennox-Gestaut syndrome, or any other seizure disorder should consult with their pediatricians closely to determine whether a cannabis-based treatment could be effective.
Finding The Right Medical Marijuana Products For You
Kind Meds is Arizona’s highest quality medical marijuana dispensary. We offer a wide range of medical cannabis products to Arizona medical marijuana cardholders with qualifying conditions. Finding the right cannabis product for you can be daunting when you see the sheer scope of variety available, and our budtenders can help you understand your options better.
If you have questions about your cannabis medication or want to learn more about Epidiolex, your first stop should be your attending physician. For all other medical marijuana needs in Arizona, come to Kind Meds.
Learn More About Different Strains in Our Strain Guide

Chris Weatherall is a cannabis researcher and content contributor focused on cannabis genetics, product education, and industry trends. His work centers on helping consumers better understand flower, concentrates, and emerging cannabis products through clear, research-driven content.